 The documented risks of these drugs are provided so the public can make informed, educated decisions. Sarafem is a newer antidepressant drug. The U.S. Food and Drug Administration warns that antidepressants can cause suicidal thinking and behavior in children and young adults. Sarafem is also known as Fluctin, Fluctine, Fluox, Fluoxetina, Fluoxetine, Fontex, Ladose, Lovan, Prozac Weekly, Verotina, Zactin.
The documented risks of these drugs are provided so the public can make informed, educated decisions. Sarafem is a newer antidepressant drug. The U.S. Food and Drug Administration warns that antidepressants can cause suicidal thinking and behavior in children and young adults. Sarafem is also known as Fluctin, Fluctine, Fluox, Fluoxetina, Fluoxetine, Fontex, Ladose, Lovan, Prozac Weekly, Verotina, Zactin.
To see the total figures from IQVia on the number of people taking antidepressants in the U.S., click here.
Please note: No one should attempt to get off of psychiatric drugs without a doctor’s supervision. To help find medical practitioners in your area, click here.
Also, read the FDA’s Sarafem Medication Guide for more information. MedGuides include “the particular serious and significant public health concern that has created the need for the Medication Guide” and notes “pediatric risks.” (Note: Unfortunately, the FDA MedGuides only work on a desktop, not on a mobile device. Please complain to the FDA to make their public advisories accessible to all. 1-888-INFO-FDA or 1-888-463-6332.)
Sarafem Drug Warnings:
There have been 28 drug regulatory agency warnings from six countries (Australia, Canada, Ireland, New Zealand, United Kingdom and United States) and the European Union on Sarafem (or fluoxetine). These include the following (note that some warnings cite more than one side effect, so the list below may not be equal to the total number of warnings):
10 warnings on Sarafem causing suicide risk or suicidal behavior
10 warnings on Sarafem causing newborn complications
8 warnings on Sarafem causing birth defects
4 warnings on Sarafem causing hostility or aggression
3 warnings on Sarafem causing agitation
3 warnings on Sarafem causing anxiety
3 warnings on Sarafem causing eye problems
2 warnings on Sarafem causing self-harm
2 warnings on Sarafem causing withdrawal reactions
2 warnings on Sarafem causing involuntary movements
2 warnings on Sarafem causing nervous system disorders
2 warnings on Sarafem causing abnormal behavior
2 warnings on Sarafem causing infant withdrawal symptoms
2 warnings on Sarafem causing violence
2 warnings on Sarafem causing abnormal bleeding
2 warnings on Sarafem causing cardiovascular disorders
1 warning on Sarafem causing Serotonin Syndrome
1 warning on Sarafem causing mania or psychosis
1 warning on Sarafem causing sleep problems
1 warning on Sarafem causing irritability
1 warning on Sarafem causing blood disorders
1 warning on Sarafem causing pregnancy complications
1 warning on Sarafem causing birth complications
1 warning on Sarafem causing premature birth
1 warning on Sarafem causing heart problems
1 warning on Sarafem causing blood pressure changes or high or low blood pressure
1 warning on Sarafem causing risk of falls
Sarafem Drug Studies:
There have been 72 studies done in 19 countries (Australia, Canada, Denmark, Finland, France, Germany, Iceland, Iran, Israel, Italy, Netherlands, New Zealand, Norway, Portugal, Spain, Sweden, Taiwan, United Kingdom and United States) on Sarafem (or fluoxetine). These include the following (note that some studies cite more than one side effect, so the list below may not be equal to the total number of studies):
17 studies on Sarafem causing suicide risk or attempts
16 studies on Sarafem causing newborn complications
7 studies on Sarafem causing abnormal bleeding
6 studies on Sarafem causing birth defects
6 studies on Sarafem causing sexual dysfunction
5 studies on Sarafem causing gastrointestinal problems
4 studies on Sarafem causing withdrawal reactions
4 studies on Sarafem causing pregnancy complications
4 studies on Sarafem causing risk of fractures
4 studies on Sarafem causing premature birth
4 studies on Sarafem causing self-harm
3 studies on Sarafem causing birth complications
3 studies on Sarafem causing nervous system disorders
3 studies on Sarafem causing involuntary movements
2 studies on Sarafem causing skin conditions
2 studies on Sarafem causing developmental disorders
2 studies on Sarafem causing hostility or aggression
2 studies on Sarafem causing infant withdrawal symptoms
1 study on Sarafem causing lack of efficacy
1 study on Sarafem causing risk of falls
1 study on Sarafem causing violence
1 study on Sarafem causing blood disorders
1 study on Sarafem causing weight gain
1 study on Sarafem causing reproductive system disorders
1 study on Sarafem causing diabetes
1 study on Sarafem causing homicide or homicidal ideation
1 study on Sarafem causing liver problems
1 study on Sarafem causing allergic reactions
1 study on Sarafem causing heart problems
1 study on Sarafem causing stroke
1 study on Sarafem causing death or increased risk of death
1 study on Sarafem causing heart attacks
1 study on Sarafem causing seizures
1 study on Sarafem causing eye problems
1 study on Sarafem causing depression
1 study on Sarafem causing mania or psychosis
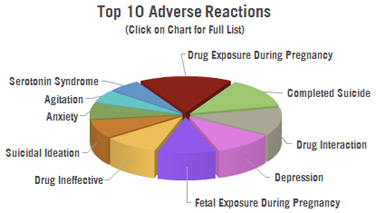
Top Reactions for All Ages: There have been 6,510 adverse reactions reported to the US FDA in connection with Sarafem.
The FDA estimates that less than 1% of all serious events are ever reported to it, so the actual number of side effects occurring are most certainly higher.
- 504 cases of completed suicides
- 407 cases of depression
- 369 cases of the drug being ineffective
- 311 cases of suicidal ideation
- 271 cases of anxiety
- 220 cases of agitation
- 220 cases of serotonin syndrome
- 204 cases of the condition being aggravated
- 191 cases of nausea
- 190 cases of feeling abnormal
Documented Side Effects of Sarafem:
Source: Physicians Desk Reference, National Institutes of Health’s Medline Plus, and/or the drug label.
Abnormal dreams
Anxiety
Difficulty breathing or swallowing
Hives
Hallucinations
Irregular heartbeat
Nervousness
Seizures
Severe muscle stiffness
Sexual dysfunction
Somnolence
Sore throat
Swelling of the face, hands, feet, or lower legs
Uncontrollable shaking of a part of the body
Weight loss
Click here to learn more >>
This brochure is a simple guide that documents the dangerous and deadly side effects of the drugs prescribed to millions of men, women and children diagnosed with bogus mental disorders.


 Download The Side Effects of Common
Download The Side Effects of Common
SHARE YOUR STORY/COMMENT