 The documented risks of these drugs are provided so the public can make informed, educated decisions. Vyvanse is a stimulant drug, classified by the U.S. Drug Enforcement Administration (DEA) as Schedule II, in the same class of highly addictive drugs as morphine, opium and cocaine. The DEA states that the use of stimulants can lead to “severe psychological or physical dependence” and that “these drugs are also considered dangerous.” Vyvanse is also known as Lisdexamfetamine.
The documented risks of these drugs are provided so the public can make informed, educated decisions. Vyvanse is a stimulant drug, classified by the U.S. Drug Enforcement Administration (DEA) as Schedule II, in the same class of highly addictive drugs as morphine, opium and cocaine. The DEA states that the use of stimulants can lead to “severe psychological or physical dependence” and that “these drugs are also considered dangerous.” Vyvanse is also known as Lisdexamfetamine.
To see the total figures from IQVia on the number of people taking stimulants in the U.S., click here.
Please note: No one should attempt to get off of psychiatric drugs without a doctor’s supervision. To help find medical practitioners in your area, click here.
Also, read the FDA’s Vyvanse Medication Guide for more information. MedGuides include “the particular serious and significant public health concern that has created the need for the Medication Guide” and notes “pediatric risks.” (Note: Unfortunately, the FDA MedGuides only work on a desktop, not on a mobile device. Please complain to the FDA to make their public advisories accessible to all. 1-888-INFO-FDA or 1-888-463-6332.)
Vyvanse Drug Warnings:
There have been nine drug regulatory agency warnings from three countries (Canada, Denmark and United States) on Vyvanse (or lisdexamfetamine). These include the following (note that some warnings cite more than one side effect, so the list below may not be equal to the total number of warnings):
2 warnings on Vyvanse causing cardiovascular disorders
1 warning on Vyvanse causing skin conditions
1 warning on Vyvanse causing suicide risk or attempts
1 warning on Vyvanse causing heart problems
1 warning on Vyvanse causing death or sudden death
1 warning on Vyvanse causing sleep problems
Vyvanse Drug Studies:
There have been three studies done in two countries (Scotland and United States) on Vyvanse (or lisdexamfetamine). These include the following (note that some studies cite more than one side effect, so the list below may not be equal to the total number of studies):
1 study on Vyvanse having lack of efficacy
1 study on Vyvanse causing stunted growth
1 study on Vyvanse causing lowered bone mass
1 study on Vyvanse causing risk of injury
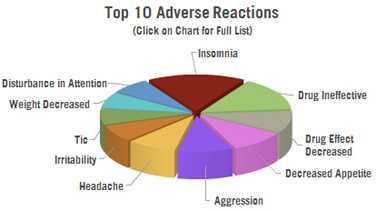
Adverse Reaction Reports Filed with the US FDA: There have been 2,991 adverse reactions reported to the US FDA in connection with Vyvanse.
The FDA estimates that less than 1% of all serious events are ever reported to it, so the actual number of side effects occurring are most certainly higher.
- 314 cases of insomnia
- 267 cases of the drug being ineffective
- 179 cases of decreased appetite
- 177 cases of aggression
- 160 cases of headache
- 140 cases of irritability
- 129 cases of tics
- 127 cases of weight decreased
- 126 cases of disturbance in attention
- 124 cases of nausea
Documented Side Effects of Vyvanse:
Source: Physicians Desk Reference, National Institutes of Health’s Medline Plus, and/or the drug label.
Aggression
Anxiety
Fainting
Fast/pounding heartbeat
Fever
Frenzied, abnormally excited mood
Hallucinations
Hives
Insomnia
Irritability
Jitters
Loss of appetite
Mood swings
Nausea
Seizures
Sleepiness
Slow or difficult speech
Sweating
Swelling of the eyes, face, tongue, lips, or mouth
Tics
Uncontrollable shaking of a part of the body
Weight decreased
Weight loss
Click here to learn more >>
This brochure is a simple guide that documents the dangerous and deadly side effects of the drugs prescribed to millions of men, women and children diagnosed with bogus mental disorders.


 Download The Side Effects of Common
Download The Side Effects of Common
SHARE YOUR STORY/COMMENT